Advertisements
Advertisements
प्रश्न
Why is sulphuric acid known as king of chemicals and oil vitriol ?
उत्तर
Sulphuric acid is known as king of chemicals because in almost all industries it is used directly or indirectly.
In 8th century it was obtained by distillation of green vitriol (FeSO4.7H2O). It is called oil of green vitriol because of its oily appearance and because of the fact that it was present in vitreous or glassy substances like ferrous sulphate etc.
APPEARS IN
संबंधित प्रश्न
State one relevant observation for given reactions:
Addition of ethyl alcohol to acetic acid in the presence of concentrated Sulphuric acid
State one relevant observation for given reactions:
Action of concentrated Sulphuric acid on hydrated copper sulfate
Which property of sulphuric acid is shown by the reaction of the concentrated sulphuric acid with Carbon?
Name the products formed when hot and concentrated sulphuric acid reacts with Sulphur.
Name the Following:
The property used to prepare HCl and HNO3 from H2SO4.
What would you observe in the following case?
Hot concentrated sulphuric acid is added to sodium chloride crystals.
Give reason for the following:
When concentrated sulphuric acid is added to sugar/glucose, a black mass is left behind.
Describe the reaction that show
Concentrated sulphuric acid is a non-volatile acid.
Give reason for the following:
Concentrated sulphuric acid is kept in airtight bottles.
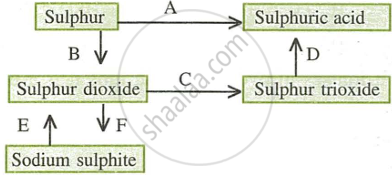
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
