Advertisements
Advertisements
प्रश्न
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
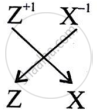
What is the formula of the compound formed between X and Z.
उत्तर
XZ is the formula of the compound formed between X and Z.
APPEARS IN
संबंधित प्रश्न
Give electron dot diagram of the following:
nitrogen
Complete the statement by filling the gaps using appropriate term from the terms given in the bracket.
(slow, coloured, arrow, fast, smell, milky, physical, product, chemical, reactant, covalent, ionic, octet, duplet, exchange, sharing, equality sign)
Sodium chloride is ______ compound while hydrogen chloride is ______ compound.
Explain the following briefly:
Helium does not form He2 molecule
A Compound 'X' consists of only molecules. Hence 'X' will have
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of oxidation.
Give the orbital diagram of the following:
Magnesium chloride
State the type of bonding in the following molecules.
Calcium oxide
Match the following:
| 1. | Monoatomic gaseous atom | a. | Electrovalent bond |
| 2. | Octet rule | b. | Benzene |
| 3. | Ionic bond | c. | Water |
| 4. | Non-polar solvent | d. | Electronic theory of valence |
| 5. | Polar solvent | e. | Noble gases |
How many atoms of each kind are present in the following molecule:
water
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is a metal.
