Advertisements
Advertisements
प्रश्न
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2, 8, 1 |
2, 8, 7 |
2, 5 |
1 |
Answer the following question based on the table above:
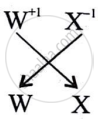
What is the formula of the compound formed between W and X.
उत्तर
WX is the formula of the compound formed between W and X.
APPEARS IN
संबंधित प्रश्न
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is a non-metal.
State the following is a reduction reaction or oxidation reaction.
\[\ce{A^3+ + e- -> A^2+}\]
Explain fractional atomic mass. What is the fractional mass of chlorine?
Choose the correct option
Which of the following is the correct electronic configuration of potassium?
Explain
Octet rule for the formation of sodium chloride
How do atoms attain noble gas configurations?
State the type of bond between Y and Z and give its molecular formula.
Explain Octet rule with an example.
How many electrons are required or released by each atom in water molecule to attain the nearest noble gas configuration?
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is a metal.
