Advertisements
Advertisements
प्रश्न
The graphs below show the variation of the stopping potential VS with the frequency (ν) of the incident radiations for two different photosensitive materials M1 and M2.
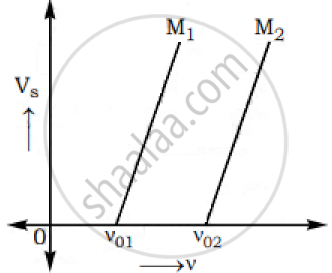
Express work function for M1 and M2 in terms of Planck’s constant(h) and Threshold frequency and charge of the electron (e).
If the values of stopping potential for M1 and M2 are V1 and V2 respectively then show that the slope of the lines equals to `(V_1-V_2)/(V_(01)-V_(02))` for a frequency,
ν > ν02 and also ν > ν01
उत्तर
`"W"_(01) = ("h"ν_(01))/"e" and "W"_(02) = ("h"ν_(02))/"e"`
`"V"_1 = ["h"/"e"] ν-["h"/"e"]"V"_01`
`"V"_2 = ["h"/"e"]ν-["h"/"e"]"V"_(02)`
∴ `"V"1 - "V"2 = ["h"/"e"]("V"_(01)-"V"_(02))`
`["h"/"e"] = ("V"1-"V"2)/("V"_(01)-"V"_(02))`
APPEARS IN
संबंधित प्रश्न
The photoelectric threshold wavelength of a metal is 230 nm. Determine the maximum kinetic energy in joule and in eV of the ejects electron for the metal surface when it is exposed to a radiation of wavelength 180 nm.
[Planck’s constant : h = 6.63 * 10-34 Js, Velocity of light : C = 3 * 108 m/s.]
Find the wave number of a photon having energy of 2.072 eV
Given : Charge on electron = 1.6 x 10-19 C,
Velocity of light in air = 3 x 108 m/s,
Planck’s constant = 6.63 x 10-34 J-s.
Define (i) stopping potential and (ii) threshold frequency, using Einstein’s equation and drawing necessary plot between relevant quantities.
Write Einstein's photoelectric equation and mention which important features in photoelectric effect can be explained with the help of this equation.
The maximum kinetic energy of the photoelectrons gets doubled when the wavelength of light incident on the surface changes from λ1 to λ2. Derive the expressions for the threshold wavelength λ0 and work function for the metal surface.
Calculate the maximum kinetic energy of photoelectrons emitted by a metal (work function = 1.5 eV) when it is illuminated with light of wavelength 198 nm.
Who indirectly determined the mass of the electron by measuring the charge of the electrons?
If the frequency of light in a photoelectric experiment is double the stopping potential will
What is the momentum of photon of energy 3 mev in kg ms-1?
The emission of electron is possible
The wavelength of matter is independent of
