Advertisements
Advertisements
प्रश्न
The photoelectric threshold wavelength of a metal is 230 nm. Determine the maximum kinetic energy in joule and in eV of the ejects electron for the metal surface when it is exposed to a radiation of wavelength 180 nm.
[Planck’s constant : h = 6.63 * 10-34 Js, Velocity of light : C = 3 * 108 m/s.]
उत्तर
λ0 = 230 nm = 230 × 10−9m,
λ = 180 nm = 180 × 10−9m,
h = 6.63 × 10−34 J s, c = 3 × 108
m/s, K.E. (in joule and eV) = ?
Now,

∴ The maximum kinetic energy of the ejected electron is 2.402 × 10−19 J.

∴ The maximum kinetic energy of the ejected electron is 1.5 eV.
APPEARS IN
संबंधित प्रश्न
The energy of photon of wavelength X is_____ .
[h = Planck’s constant, c = speed of light in vacuum]
The momentum of a photon of de Broglie wavelength 5000Å is _______.
[Planck’s constant = 6.63 x10-34 J.s.]
When radiations of wavelength λ1 and λ2 are incident on certain photosensitive, such that E1 > E2 . Then Planck's constant 'h' is ......................... .
(C = Velocity of light).
Find the value of energy of electron in eV in the third Bohr orbit of hydrogen atom.
(Rydberg's constant (R) = 1· 097 x 107m - 1,Planck's constant (h) =6·63x10-34 J-s,Velocity of light in air (c) = 3 x 108m/ s.)
State two important properties of photon which are used to write Einstein’s photoelectric equation.
Define (i) stopping potential and (ii) threshold frequency, using Einstein’s equation and drawing necessary plot between relevant quantities.
A proton and a deuteron are accelerated through the same accelerating potential. Which one of the two has less momentum?
Give reasons to justify your answer.
Radiation of frequency 1015 Hz is incident on two photosensitive surface P and Q. There is no photoemission from surface P. Photoemission occurs from surface Q but photoelectrons have zero kinetic energy. Explain these observations and find the value of work function for surface Q.
Einstein's photoelectric equation is:
a) `E_"max" = hlambda - varphi_0`
b) `E_"max"= (hc)/lambda varphi_0`
c) `E_"max" = hv + varphi_0`
d) `E_"max" = (hv)/lambda + varphi_0`
With reference to the photoelectric effect, define threshold wavelength
Write the basic features of photon picture of electromagnetic radiation on which Einstein’s photoelectric equation is based.
Write Einstein’s photoelectric equation.
According to Einstein’s model minimum energy needed for the electron to escape from a metal surface having work function ϕ0, the electron is emitted with maximum kinetic energy, Kmax = ______.
According to Einstein’s model, the threshold frequency for a metal having work function ϕ0 is given by _________.
Radiations of two photon's energy, twice and ten times the work function of metal are incident on the metal surface successively. The ratio of maximum velocities of photoelectrons emitted in two cases is:
A 200 W sodium street lamp emits yellow light of wavelength 0.6 µm. Assuming it to be 25% efficient in converting electrical energy to light, the number of photons of yellow light it emits per second is:
Dimensions of ‘resistance’ are same as (where h is Planck's constant and e is charge):
Threshold frequency for photoelectric effect on sodium corresponds to a wave length 5000. Its work function is ______.
If the frequency of light in a photoelectric experiment is double the stopping potential will
If the energy of photon corresponding to a wavelength of 6000 A° is 3.32 × 10-19 J, the photon energy for a wavelength of 4000 A° will be
The slope of frequency of incident light and stopping potential for a given surface will be
What will be wavelength of a photon of momentum 6.6 × 10–24 kgms–1?
The emission of electron is possible
(i) A ray of light incident on face AB of an equilateral glass prism, shows minimum deviation of 30°. Calculate the speed of light through the prism.
(ii) Find the angle of incidence at face AB so that the emergent ray grazes along the face AC.
- Calculate the energy and momentum of a photon in a monochromatic beam of wavelength 331.5 nm.
- How fast should a hydrogen atom travel in order to have the same momentum as that of the photon in part (a)?
- Calculate the frequency of a photon of energy 6.5 × 10−19 J.
- Can this photon cause the emission of an electron from the surface of Cs of work function 2.14 eV? If yes, what will be the maximum kinetic energy of the photoelectron?
How does stopping potential in photoelectric emission vary if the intensity of the incident radiation increases?
The graphs below show the variation of the stopping potential VS with the frequency (ν) of the incident radiations for two different photosensitive materials M1 and M2.
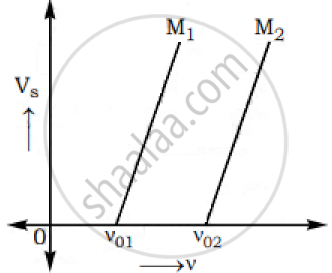
Express work function for M1 and M2 in terms of Planck’s constant(h) and Threshold frequency and charge of the electron (e).
If the values of stopping potential for M1 and M2 are V1 and V2 respectively then show that the slope of the lines equals to `(V_1-V_2)/(V_(01)-V_(02))` for a frequency,
ν > ν02 and also ν > ν01
