Advertisements
Advertisements
प्रश्न
The slope in the plot of ln[R] vs. time for a first order reaction is ______.
पर्याय
`(+k)/2.303`
– k
`(-k)/2.303`
+ k
उत्तर
The slope in the plot of ln[R] vs. time for a first order reaction is – k.
Explanation:
For the reaction, R `rightarrow` Products
The integrated first order rate equation is
ln [R] = – kt + ln [R0]
Comparing the above equation with a straight line equation y = mx + c
∴ y = ln [R], x = t, slope = – k and intercept = ln [R0]
Thus, the slope for first order reaction is – k.
APPEARS IN
संबंधित प्रश्न
A first order reaction has a rate constant 1.15 × 10−3 s−1. How long will 5 g of this reactant take to reduce to 3 g?
The rate constant for a first order reaction is 60 s−1. How much time will it take to reduce the initial concentration of the reactant to its `1/16`th value?
Define order of reaction. How does order of a reaction differ from molecularity for a complex reaction?
Which of the following graphs is correct for a first order reaction?

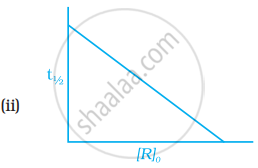
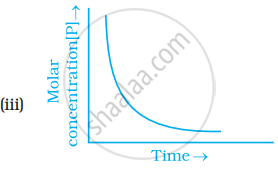
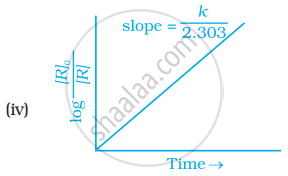
The rate constant of a first order reaction is 6.9 × 10–3s–1. How much time will it take to reduce the initial concentration to its 1/8th value?
The decomposition of formic acid on gold surface follows first-order kinetics. If the rate constant at 300 K is 1.0 × 10−3 s−1 and the activation energy Ea = 11.488 kJ mol−1, the rate constant at 200 K is ______ × 10−5 s−1. (Round off to the Nearest Integer)
(Given R = 8.314 J mol−1 K−1)
How will you represent first order reactions graphically?
Define first-order reaction.
Write the equation for integrated rate law for a first order reaction.
Write the unit of rate constant [k] for the first order reaction.
