Advertisements
Advertisements
प्रश्न
The types of hybrid orbitals of nitrogen in \[\ce{NO^{+}2}\] , \[\ce{NO^{-}3}\] and \[\ce{NH^{+}4}\] respectively are expected to be ______.
पर्याय
sp, sp3 and sp2
sp, sp2 and sp3
sp2, sp and sp3
sp2, sp3 and sp
उत्तर
The types of hybrid orbitals of nitrogen in \[\ce{NO^{+}2}\] , \[\ce{NO^{-}3}\] and \[\ce{NH^{+}4}\] respectively are expected to be sp, sp2 and sp3.
Explanation:
The number of orbitals involved in hybridization can be determined by the application of formula:
\[\ce{H = 1/2 [V + M - C + A]}\]
Where H = number of orbitals involved in hybridization
V = valence electrons of central atom
M = number of monovalent atoms linked with central atom
C = charge on the cation
A = charge on the anion
\[\ce{NO^{+}2 : H = 1/2 [5 + 0 - 1 + 0] = 2 or sp}\]
\[\ce{NO^{-}3 : H = 1/2 [5 + 0 - 0 + 1] = 3 or sp^2}\]
\[\ce{NH^{+}4 : H = 1/2 [5 + 4 - 1 + 0] = 4 or sp^3}\]
APPEARS IN
संबंधित प्रश्न
What is the total number of sigma and pi bonds in the following molecules?
C2H4
Isostructural species are those which have the same shape and hybridisation. Among the given species identify the isostructural pairs.
Discuss the concept of hybridisation. What are its different types in a carbon atom.
What is the type of hybridisation of carbon atoms marked with star.
\[\begin{array}{cc}
\phantom{.....}\ce{O}\\
\phantom{.....}||\\
\ce{\overset{∗}{C}H2 = CH - \overset{∗}{C} - O - H}
\end{array}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C}H2 - OH}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{\overset{∗}{C}H3 - CH = CH - CH3}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C} ≡ CH}\]
BF3 is a planar and electron-deficient compound. Hybridization and the number of electrons around the central atom, respectively are ______.
In the given reaction,
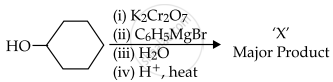
the number of sp2 hybridised carbon (s) in compound 'X' is ______.
In which of the following species S atom assumes sp3 hybrid state?
(I) (SO3)
(II) SO2
(III) H2S
(IV) S8
