Advertisements
Advertisements
Question
The types of hybrid orbitals of nitrogen in \[\ce{NO^{+}2}\] , \[\ce{NO^{-}3}\] and \[\ce{NH^{+}4}\] respectively are expected to be ______.
Options
sp, sp3 and sp2
sp, sp2 and sp3
sp2, sp and sp3
sp2, sp3 and sp
Solution
The types of hybrid orbitals of nitrogen in \[\ce{NO^{+}2}\] , \[\ce{NO^{-}3}\] and \[\ce{NH^{+}4}\] respectively are expected to be sp, sp2 and sp3.
Explanation:
The number of orbitals involved in hybridization can be determined by the application of formula:
\[\ce{H = 1/2 [V + M - C + A]}\]
Where H = number of orbitals involved in hybridization
V = valence electrons of central atom
M = number of monovalent atoms linked with central atom
C = charge on the cation
A = charge on the anion
\[\ce{NO^{+}2 : H = 1/2 [5 + 0 - 1 + 0] = 2 or sp}\]
\[\ce{NO^{-}3 : H = 1/2 [5 + 0 - 0 + 1] = 3 or sp^2}\]
\[\ce{NH^{+}4 : H = 1/2 [5 + 4 - 1 + 0] = 4 or sp^3}\]
APPEARS IN
RELATED QUESTIONS
What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type.
Predict the shapes of the following molecules on the basis of hybridisation.
\[\ce{BCl3, CH4 , CO2, NH3}\]
Match the species in Column I with the type of hybrid orbitals in Column II.
| Column I | Column II |
| (i) \[\ce{SF4}\] | (a) sp3d2 |
| (ii) \[\ce{IF5}\] | (b) d2sp3 |
| (iii) \[\ce{NO^{+}2}\], | (c) sp3d |
| (iv) \[\ce{NH^{+}4}\], | (d) sp3 |
| (e) sp |
Match the shape of molecules in Column I with the type of hybridisation in Column II.
| Column I | Column II |
| (i) Tetrahedral | (a) sp2 |
| (ii) Trigonal | (b) sp |
| (iii) Linear | (c) sp3 |
Discuss the concept of hybridisation. What are its different types in a carbon atom.
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C}H2 - OH}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{\overset{∗}{C}H3 - CH = CH - CH3}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C} ≡ CH}\]
In the given reaction,
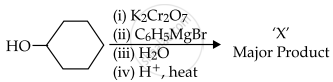
the number of sp2 hybridised carbon (s) in compound 'X' is ______.
The hybridisation of carbanion is:
