Advertisements
Advertisements
Question
Predict the shapes of the following molecules on the basis of hybridisation.
\[\ce{BCl3, CH4 , CO2, NH3}\]
Solution
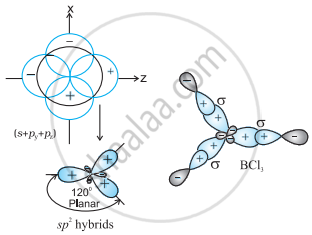
In \[\ce{BCl3}\], the geometry is trigonal planar is due to sp2 hybridisation.
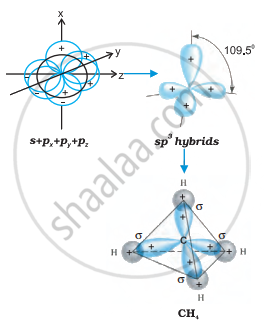
The shape of \[\ce{CH4}\] is tetrahedral due to sp3 hybridisation.
\[\ce{CO2}\] show linear shape because of sp hybridisation.

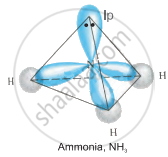
The geometry of \[\ce{NH3}\] is pyramidal shape and has sp3 hybridisation.
APPEARS IN
RELATED QUESTIONS
Apart from tetrahedral geometry, another possible geometry for CH4 is square planar with the four H atoms at the corners of the square and the C atom at its centre. Explain why CH4 is not square planar?
Distinguish between a sigma and a pi bond.
Isostructural species are those which have the same shape and hybridisation. Among the given species identify the isostructural pairs.
The types of hybrid orbitals of nitrogen in \[\ce{NO^{+}2}\] , \[\ce{NO^{-}3}\] and \[\ce{NH^{+}4}\] respectively are expected to be ______.
Which of the following angle corresponds to sp2 hybridisation?
Match the shape of molecules in Column I with the type of hybridisation in Column II.
| Column I | Column II |
| (i) Tetrahedral | (a) sp2 |
| (ii) Trigonal | (b) sp |
| (iii) Linear | (c) sp3 |
What is the type of hybridisation of carbon atoms marked with star.
\[\begin{array}{cc}
\phantom{.....}\ce{O}\\
\phantom{.....}||\\
\ce{\overset{∗}{C}H2 = CH - \overset{∗}{C} - O - H}
\end{array}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{\overset{∗}{C}H3 - CH = CH - CH3}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C} ≡ CH}\]
In the given reaction,
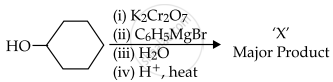
the number of sp2 hybridised carbon (s) in compound 'X' is ______.
