Advertisements
Advertisements
Question
Hydrogen bonds are formed in many compounds e.g., \[\ce{H2O, HF, NH3}\]. The boiling point of such compounds depends to a large extent on the strength of hydrogen bond and the number of hydrogen bonds. The correct decreasing order of the boiling points of above compounds is ______.
Options
\[\ce{HF > H2O > NH3}\]
\[\ce{H2O > HF > NH3}\]
\[\ce{NH3 > HF > H2O}\]
\[\ce{NH3 > H2O > HF}\]
Solution
Hydrogen bonds are formed in many compounds e.g., \[\ce{H2O, HF, NH3}\]. The boiling point of such compounds depends to a large extent on the strength of hydrogen bond and the number of hydrogen bonds. The correct decreasing order of the boiling points of above compounds is \[\ce{H2O > HF > NH3}\].
Explanation:
Strength of H-bonding depends on the electronegativity of the atom which follows the order: \[\ce{F > O > N}\].
Strength of H-bond is in the order:
\[\ce{H}\]……. \[\ce{F > H}\]…….. \[\ce{O > H}\]…….. \[\ce{N}\]
But each \[\ce{H2O}\] molecule is linked to 4 other \[\ce{H2O}\] molecules through H-bonds whereas each \[\ce{HF}\] molecule is linked only to two other \[\ce{HF}\] molecules.
APPEARS IN
RELATED QUESTIONS
In which of the following substances will hydrogen bond be strongest?
Structures of molecules of two compounds are given below :
 |
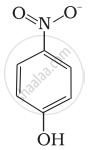 |
| (I) | (II) |
- Which of the two compounds will have intermolecular hydrogen bonding and which compound is expected to show intramolecular hydrogen bonding.
- The melting point of a compound depends on, among other things, the extent of hydrogen bonding. On this basis explain which of the above two compounds will show higher melting point.
- Solubility of compounds in water depends on power to form hydrogen bonds with water. Which of the above compounds will form hydrogen bond with water easily and be more soluble in it.
The boiling point of methanol is greater than of methylthiol because ______.
Assertion A: Enol form of acetone [CH3COCH3] exists in < 0.1% quantity. However, the enol form the acetyl acetone [CH3COCH2OCCH3] exists in approximately 15% quantity.
Reason R: Enol form of acetyl acetone is stabilized by intramolecular hydrogen bonding, which is not possible in enol form of acetone.
Choose the correct answer:
