Advertisements
Advertisements
प्रश्न
The variation of pressure with volume of the gas at different temperatures can be graphically represented as shown in figure.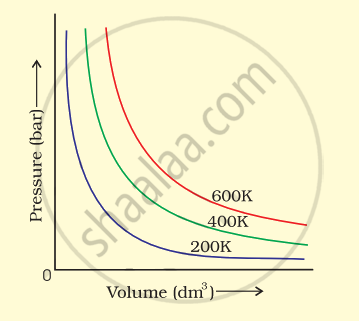
On the basis of this graph answer the following questions.
(i) How will the volume of a gas change if its pressure is increased at constant temperature?
(ii) At a constant pressure, how will the volume of a gas change if the temperature is increased from 200 K to 400 K?
उत्तर
(i) Pressure of a gas is inversely proportional to volume of gas at constant temperature. So, the volume decreases with increase in pressure at constant temperature.
(ii) Volume of a gas is directly proportional to temperature when the pressure is constant. So, the volume of gas increases with increase in temperature.
APPEARS IN
संबंधित प्रश्न
A student forgot to add the reaction mixture to the round-bottomed flask at 27 °C but instead, he/she placed the flask on the flame. After a lapse of time, he realized his mistake and using a pyrometer he found the temperature of the flask was 477 °C. What fraction of air would have been expelled out?
In terms of Charles’ law explain why - 273°C is the lowest possible temperature.
Match the following gas laws with the equation representing them.
| (i) Boyle's law | (a) `V oo n` at constant T and p |
| (ii) Charle's law | (b) PTotal = p1 + p2 + p3 + at constant T, V |
| (iii) Dalton's law | (c) `(pV)/T` = Constant T |
| (iv) Avogadro law | (d) `V oo T` at constant n and p |
| (e) `P oo 1/V` at constant n and T |
If V0 is the volume of a given mass of gas at 273 K at a constant pressure then according to Charle's law, the volume at 10°C will be:
