Advertisements
Advertisements
प्रश्न
What is dipole moment?
उत्तर
The polarity of a covalent bond can be measured in terms of dipole moment which is defined as
μ = q × 2d
Where μ is the dipole moment, q is the charge and 2d is the distance between the two charges. The dipole moment is a vector and the direction of the dipole moment vector points from the negative charge to positive charge.
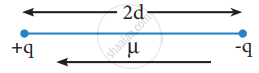
Representation of dipole
The unit of dipole moment is coloumb meter (C m). It is usually expressed in Debye unit (d). The conversion factor is 1 Debye = 3.336 × 10-30 C m.
APPEARS IN
संबंधित प्रश्न
Non – Zero dipole moment is shown by ______.
Define bond order.
Define bond energy.
Explain resonance with reference to a carbonate ion.
Explain the bond formation in ethylene.
Explain the bond formation in acetylene.
Which one of the following has the highest bond order?
- N2
- N2+
- N2–
Describe Fajan’s rule.
The bond order in NO is 2.5 while that in NO+ is 3. Which of the following statements is true for these two species?
Bond distance in HF is 9.17 × 10−11 m. Dipole moment of HF is 6.104 × 10−30 Cm. The percentage of ionic character in HF will be ______.
(electron charge = 1.60 × 10−19 C)
