Advertisements
Advertisements
प्रश्न
What is meant by refining of a metal? Name the most widely used method for the refining of impure metals obtained by various reduction processes. Describe this method with the help of a labelled diagram by taking the example of any metal.
उत्तर
The metals obtained by different reduction processes are usually found to be with some impurities. The process involved in the removal of these impurities from the reduced metals is called purification or refining of metals. There are several refining methods used to purify metals based on the type of metals.
The most widely used method for the refining of impure metals obtained by various reduction processes is electrolytic refining method. Electrolytic refining is a process of purification by electrolysis. Several metals like copper, zinc, tin, lead, nickel, chromium, gold and silver are refined by this process.
Let us explain electrolytic refining process by considering an example of copper metal. The electrolytic refining tank consists of anode, cathode and electrolytic solution. In the refining of copper metal, a bulky piece of impure copper is made anode (attached to positive terminal) and a thin layer of pure copper is made cathode (attached to negative terminal). The acidified copper sulphate solution (copper sulphate solution with dilute sulphuric acid) is taken as electrolytic solution or electrolyte.

When electricity is passed through the cell, impure copper from the anode (as CU2+ ions) melts and travels to the electrolyte. The pure copper metal (CU2+ ions) from the electrolyte moves to the cathode and gets deposited there as pure copper. The amount of copper ions from the anode going to the electrolyte will be equivalent to the amount of copper ions collected at the cathode. This keeps the concentration of the electrolyte constant and proceeds the further reaction. Copper ions, being positively charged, get attracted to the negatively charged cathode and are collected or deposited there.
The copper sulphate electrolyte contains CU2+ and `SO_4^(2-)` ions. The reactions that take place at anode and cathode when electricity passed through the electrolytic cell are:
(i) The impure copper atoms from anode lose two electrons to form copper ions and enter the copper sulphate electrolyte.
Anode: 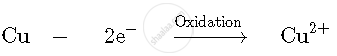
(ii) The copper ions from the copper sulphate electrolyte move to cathode (negatively charged) by gaining two electrons from cathode and get reduced to copper atoms.
Cathode: 
Thus impure copper atoms, entering copper sulphate electrolyte from anode, get deposited as pure copper atoms at cathode. As the process continues, cathode (pure metal collected) becomes thicker and anode (impure metal) becomes thinner. The metal obtained would be 99.99 percent pure. This pure copper is separated from the cathode later. The impurities are thrown back under the anode as anode mud.
संबंधित प्रश्न
What is aqua-regia? Name two special metals which are insoluble in common reagents but dissolve in aqua-regia.
During the refining of an impure metal by electrolysis, the pure metal is a deposited:
(a) at cathode
(b) on the walls of electrolytic tank
(c) at anode
(d) at the bottom of electrolytic tank
Explain how the following metals are obtained from their compounds by the reduction process:
(i) Metal M which is in the middle of the reactivity series.
(ii) Metal N which is high up in the reactivity series.
Give one example of each type
Which of the following process to be carried out to avoid the
formation of greenish layer on brass vessels due to corrosion?
Name one metal each occurring as :
A sulphine
In the electrolytic refining of copper, the electrolyte used is:
An electrolytic cell consists of
- positively charged cathode
- negatively charged anode
- positively charged anode
- negatively charged cathode
A reddish-brown metal ‘X’, when heated in air, gives a black compound ‘Y’, which when heated in the presence of H2 gas gives ‘X’ back. ‘X’ is refined by the process of electrolysis; this refined form of ‘X’ is used in electrical wiring. Identify ‘X’ and ‘Y’. Draw a well-labelled diagram to represent the process of refining ‘X’.
Answer the following question with reference to the electrorefining of copper:
What is the anode made of?
Answer the following question with reference to the electrorefining of copper:
What do you observe at the cathode?
