Advertisements
Advertisements
प्रश्न
Explain how the following metals are obtained from their compounds by the reduction process:
(i) Metal M which is in the middle of the reactivity series.
(ii) Metal N which is high up in the reactivity series.
Give one example of each type
उत्तर
(i) Metals that are present at the centre of the reactivity series are moderately-reactive. Therefore, metal M is a moderately-reactive metal. After obtaining the metal oxides from ores, the metal oxides are reduced to obtain the pure metals. As metal M is a moderately reactive metal, its oxide can be reduced by using coke (carbon).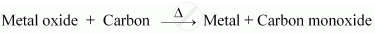
One example of metal M: Iron (Fe)
(ii) Metals that are present higher up in the reactivity series are very reactive metals. Hence, metal N is a highly reactive metal. Oxides of highly reactive metals cannot be reduced by using coke. Therefore, these metals are reduced by passing electric current throughtheir molten salts. This process is known as electrolytic reduction.
One example of metal N: Sodium (Na)
APPEARS IN
संबंधित प्रश्न
Name one method for the refining of metals.
During the refining of an impure metal by electrolysis, the pure metal is a deposited:
(a) at cathode
(b) on the walls of electrolytic tank
(c) at anode
(d) at the bottom of electrolytic tank
In the electrolytic refining of copper, the electrolyte used is:
Metals are refined by using different methods. Which of the following metals are refined by electrolytic refining?
(i) Au
(ii) Cu
(iii) Na
(iv) K
An electrolytic cell consists of
- positively charged cathode
- negatively charged anode
- positively charged anode
- negatively charged cathode
Reaction between X and Y, forms compound Z. X loses electron and Y gains electron. Which of the following properties is not shown by Z?
Which one of the following figures correctly describes the process of electrolytic refining?
What are the constituents of solder alloy? Which property of solder makes it suitable for welding electrical wires?
A reddish-brown metal ‘X’, when heated in air, gives a black compound ‘Y’, which when heated in the presence of H2 gas gives ‘X’ back. ‘X’ is refined by the process of electrolysis; this refined form of ‘X’ is used in electrical wiring. Identify ‘X’ and ‘Y’. Draw a well-labelled diagram to represent the process of refining ‘X’.
Answer the following question with reference to the electrorefining of copper:
What do you observe at the cathode?
