Advertisements
Advertisements
प्रश्न
What type of reaction is represented by the following equation?
CaCO3 → CaO + CO2
उत्तर
Decomposition reaction by the action of heat
APPEARS IN
संबंधित प्रश्न
Write one equation for decomposition reactions where energy is supplied in the form of heat.
Decomposition reactions require energy either in the form of heat or light or electricity for breaking down the reactants. Write one equation each for decomposition reactions where energy is supplied in the form of heat, light and electricity
Identify the type of following reaction :
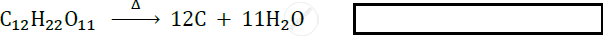
(a) Design an activity to demonstrate the decomposition reaction of lead nitrate.
(b) Draw labelled diagram of the experimental set-up. List two main observations.
(c) Write balanced chemical equation for the reaction stating the physical state of the reactant and the products.
Classify the following reaction into –
- Direct combination
- Decomposition
- Displacement
- Double decomposition
The reaction is – Zinc hydroxide on heating gives zinc oxide and water.
The chemical reaction in which two or more products are formed from a single reactant is called _______ reaction.
Chemical volcano is an example of ______ type of reaction.
Balance the following chemical equation and identify the type of chemical reaction.
`"H"_2"O"_2("l") overset("U V")(->) "H"_2"O"("l") + "O"_2("g")`
When lead nitrate is heated strongly in a boiling tube, two gases are liberated and a solid residue is left behind in the test tube.
- Name the type of chemical reaction and define it.
- Write the name and formula of the coloured gas liberated.
- Write the balanced chemical equation for the reaction.
- Name the residue left in the test tube and state the method of testing its nature (acidic/basic).
