Advertisements
Advertisements
प्रश्न
Decomposition reactions require energy either in the form of heat or light or electricity for breaking down the reactants. Write one equation each for decomposition reactions where energy is supplied in the form of heat, light and electricity
उत्तर
Those reactions in which a compound splits up into two or more simpler substances are known as decomposition reactions.
1)
![]()
2)
`2AgCl_((s))` + Sunlight  `2Ag_(s) + Cl_(2(g))`
`2Ag_(s) + Cl_(2(g))`
3)
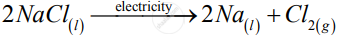
APPEARS IN
संबंधित प्रश्न
Give one example of a decomposition reaction which is carried out by applying heat.
What type of reaction is represented by the following equation?
NH4 CI → NH3 + HCI
Classify the following reaction as combination, decomposition, displacement, precipitation and neutralization. Also balance the equation.
\[\ce{Zn_{(s)} + H2SO4 -> ZnSO4_{(s)} + H2_{(g)}}\]
How will you obtain Nitrogen dioxide from lead nitrate.
Also give balanced equations for the reactions
What does one mean by endothermic reaction? Give example.
Give a balanced equation for –
A thermal decomposition reaction involving heat on limestone [calcium carbonate]
The chemical reaction in which two or more products are formed from a single reactant is called _______ reaction.
Explain the reaction given in the figure.

Balance the following chemical equation and identify the type of chemical reaction.
`"HgO"("s") overset("(heat)")(->) "Hg"("l") + "O"_2("g")`
Write one equation for decomposition reactions where energy is supplied in the form of electricity.
