Advertisements
Advertisements
प्रश्न
Write one equation for decomposition reactions where energy is supplied in the form of electricity.
उत्तर
Decomposition reaction involving absorption of electrical energy,
- \[\ce{\underset{Alumina}{2Al2O3_{(l)}} ->[Electric current] \underset{Aluminium}{4Al_{(l)}} + \underset{Oxygen}{3O2_{(g)}}}\]
- \[\ce{2H2O_{(l)} ->[electricity] 2H2_{(g)} + O2_{(g)}}\]
संबंधित प्रश्न
Decomposition reactions require energy either in the form of heat or light or electricity for breaking down the reactants. Write one equation each for decomposition reactions where energy is supplied in the form of heat, light and electricity
What type of reaction is represented by the following equation?
CaCO3 → CaO + CO2
What type of chemical reaction take place when lime-stone is heated?
Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
A metal salt MX when exposed to light splits up to form metal M and a gas X2. Metal M is used in making ornaments whereas gas X2 is used in making bleaching powder. The salt MX is itself used in black and white photography.
(a) What do you think metal M is?
(b) What could be gas X2?
(c) Name the metal salt MX.
(d) Name any two salt solutions which on mixing together can produce a precipitate of salt MX.
(e) What type of chemical reaction takes place when salt MX is exposed to light? Write the equation of the reaction?
Explain the following type of chemical reaction, giving two examples for it:
Decomposition reaction
What do you mean by redox reaction ? Explain with the help of an example.
Identify the type of following reaction :
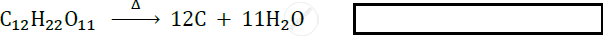
(a) Design an activity to demonstrate the decomposition reaction of lead nitrate.
(b) Draw labelled diagram of the experimental set-up. List two main observations.
(c) Write balanced chemical equation for the reaction stating the physical state of the reactant and the products.
Answer the following question.
2 g of silver chloride is taken in a china dish and the china dish is placed in sunlight for some time. What will be your observation in this case? Write the chemical reaction involved in the form of a balanced chemical equation. Identify the type of chemical reaction.
Give a balanced equation for the following type of reaction:
A thermal decomposition reaction in which a compound decomposes to give two new compounds.
Differentiate between the following:
Thermal decomposition and thermal dissociation.
Differentiate between the following:
Electrolytic decomposition and photochemical decomposition.
Electrolysis is type of ______ reaction.
Which one of the following processes involves chemical reactions?
These consist of two statements – Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
Assertion: Silver bromide decomposition is used in black and white photography.
Reason: Light provides energy for this exothermic reaction.
