Advertisements
Advertisements
प्रश्न
Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
उत्तर
(c) AgCl
The chemical reaction is given as follows:
2AgCl (s) → 2Ag (s) + Cl2 (g)
APPEARS IN
संबंधित प्रश्न
Give one example of a decomposition reaction which is carried out with electricity.
What type of chemical reaction is represented by the following equation?
X → Y + Z
MULTIPLE CHOICE QUESTIONS
Tick the most appropriate answer.
Thermal decomposition of a substance is brought about with the help of
- reactants
- water
- wind
- heat
What are thermal decomposition reactions ? Explain with an example.
Identify the type of following reaction :
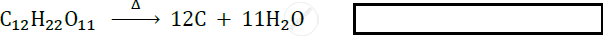
How can the rate of the chemical reaction, namely, decomposition of hydrogen peroxide be increased?
A metal nitrate 'A' on heating gives a metal oxide along with evolution of a brown coloured gas 'B' and a colourless gas, which helps in burning. Aqueous solution of 'A' when reacted with potassium iodide forms a yellow precipitate.
- Identify 'A' and 'B'
- Name the types of the reactions involved in the above statement.
What is observed when silver chloride is exposed to sunlight? Give the type of reaction involved.
Write one equation for decomposition reactions where energy is supplied in the form of electricity.
