Advertisements
Advertisements
प्रश्न
How can the rate of the chemical reaction, namely, decomposition of hydrogen peroxide be increased?
उत्तर
\[\ce{\underset{peroxide}{\underset{Hydrogen}{2H2O}_{2(l)}} -> \underset{water}{2H2O} + \underset{oxygen}{O2\uparrow}}\]
- The decomposition of hydrogen peroxide (H2O2) into water and oxygen takes place slowly at room temperature.
- However, hydrogen peroxide decomposes rapidly in the presence of manganese dioxide (MnO2) powder, which acts as a catalyst.
- Thus, by adding manganese dioxide (MnO2) powder, the rate of a chemical reaction can be increased.
APPEARS IN
संबंधित प्रश्न
Why are decomposition reactions called the opposite of combination reactions? Write equations for these reactions.
The white solid compound A decomposes quite rapidly on heating in the presence of a black substance X to form a solid compound B and a gas C. When an aqueous solution of compound B is reacted with silver nitrate solution, then a white precipitate of silver chloride is obtained along with potassium nitrate solution. Gas C does not burn itself but helps burn other things.
(a) What is compound A?
(b) What is compound B?
(c) What is gas C?
(d) What do you think is the black substance X? What is its function?
(e) What is the general name of substances like X?
Give one example of a decomposition reaction which is carried out by applying heat.
What type of chemical reaction is used to extract metals from their naturally occurring compounds like oxides or chlorides?
What type of reaction is represented by the following equation?
NH4 CI → NH3 + HCI
What type of chemical reaction take place when lime-stone is heated?
What type of chemical reaction take place when electricity is passed through water?
What type of chemical reaction is represented by the following equation?
X → Y + Z
Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
When a green iron salt is heated strongly, its colour finally changes to brown and odour of burning sulphur is given out.
(a) Name the iron salt.
(b) Name the type of reaction that takes place during the heating of iron salt.
(c) Write a chemical equation for the reaction involved.
Two metals X and Y form the salts XSO4 and Y2SO4, respectively. The solution of salt XSO4 is blue in colour whereas that of Y2SO4 is colourless. When barium chloride solution is added to XSO4 solution, then a white precipitate Z is formed alongwith a salt which turns the solution green. And when barium chloride solution is added to Y2SO4 solution, then the same white precipitate Z is formed alongwith colourless common salt solution.
(a) What could the metals X and Y be?
(b) Write the name and formula of salt XSO4.
(c) Write the name and formula of salt Y2SO4.
(d) What is the name and formula of white precipitate Z?
(e) Write the name and formula of the salt which turns the solution green in the first case.
How will you obtain Nitrogen dioxide from lead nitrate.
Also give balanced equations for the reactions
MULTIPLE CHOICE QUESTIONS
Tick the most appropriate answer.
Thermal decomposition of a substance is brought about with the help of
- reactants
- water
- wind
- heat
What do you mean by redox reaction ? Explain with the help of an example.
Identify the type of following reaction :
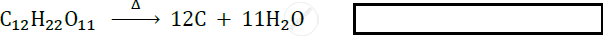
Study the following figure and answer questions.

a) After heating Calcium carbonate, which gas is formed in a test tube?
b) When we pass this gas through limewater what change, did you observe?
c) Write down the chemical reaction showing the product formation after heating the Calcium carbonate.
Classify the following reaction into different type:
\[\ce{2KClO3(s)->[\Delta] 2KCl(aq) + 3O2(g)}\]
Classify the following reaction into –
- Direct combination
- Decomposition
- Displacement
- Double decomposition
The reaction is – Zinc hydroxide on heating gives zinc oxide and water.
Give a balanced equation for –
A thermal decomposition reaction involving heat on limestone [calcium carbonate]
Give a balanced equation for –
A double decomposition neutralization reaction involving an acid and a base
Give a balanced equation for –
A white precipitate obtained during a double decomposition reaction involving a silver salt with sodium salt.
Give a balanced equation for the following type of reaction:
A thermal decomposition reaction in which a compound decomposes to give two new compounds.
The chemical reaction in which two or more products are formed from a single reactant is called _______ reaction.
Electrolysis is type of ______ reaction.
Chemical volcano is an example of ______ type of reaction.
The following reaction is used for the preparation of oxygen gas in the laboratory:
\[\ce{2KClO3_{(s)} ->[Heat] 2KCl + 3O2_{(g)}}\]
Which of the following statement about the reaction is correct?
Which among the following statement(s) is (are) true?
Exposure of silver chloride to sunlight for a long duration turns grey due to
(i) the formation of silver by decomposition of silver chloride.
(ii) sublimation of silver chloride.
(iii) decomposition of chlorine gas from silver chloride.
(iv) oxidation of silver chloride.
When SO2 gas is passed through a saturated solution of H2S, which of the following reaction occurs?
Assertion: Decomposition of vegetable matter into compost is an endothermic reaction.
Reason: Decomposition reaction involves breakdown of a single reactant into simpler products.
Complete the following reaction:
\[\ce{C_12H_22O11->[Heat]}\] ______ + ______.
A metal nitrate 'A' on heating gives a metal oxide along with evolution of a brown coloured gas 'B' and a colourless gas, which helps in burning. Aqueous solution of 'A' when reacted with potassium iodide forms a yellow precipitate.
- Identify 'A' and 'B'
- Name the types of the reactions involved in the above statement.
Write the molecular formula of calcium carbonate.
