Advertisements
Advertisements
Question
Decomposition reactions require energy either in the form of heat or light or electricity for breaking down the reactants. Write one equation each for decomposition reactions where energy is supplied in the form of heat, light and electricity
Solution
Those reactions in which a compound splits up into two or more simpler substances are known as decomposition reactions.
1)
![]()
2)
`2AgCl_((s))` + Sunlight  `2Ag_(s) + Cl_(2(g))`
`2Ag_(s) + Cl_(2(g))`
3)
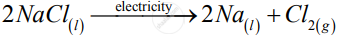
APPEARS IN
RELATED QUESTIONS
Why are decomposition reactions called the opposite of combination reactions? Write equations for these reactions.
What type of reaction is represented by the following equation?
\[\ce{CaCO3 -> CaCO + CO2}\]
What type of reaction is represented by the following equation?
NH4NO2 → N2 + 2H2O
Name the product formed on strongly heating ferrous sulphate crystals. What type of chemical reaction occurs in this change?
Give scientific reason.
When the gas formed on heating limestone, is passed through freshly prepared lime water, the lime water turns milky.
Differentiate between direct combination reaction and a decomposition reaction.
Photolysis is a decomposition reaction caused by ______
Assertion: Decomposition of vegetable matter into compost is an endothermic reaction.
Reason: Decomposition reaction involves breakdown of a single reactant into simpler products.
Balance the following chemical equation and identify the type of chemical reaction.
`"HgO"("s") overset("(heat)")(->) "Hg"("l") + "O"_2("g")`
These consist of two statements – Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
Assertion: Silver bromide decomposition is used in black and white photography.
Reason: Light provides energy for this exothermic reaction.
