Advertisements
Advertisements
प्रश्न
Which acid of the pair shown here would you expect to be stronger?
CH2FCH2CH2CO2H or CH3CHFCH2CO2H
उत्तर
| \[\begin{array}{cc} \phantom{...................}\ce{O}\\ \phantom{...................}||\\ \ce{\underset{4-Fluorobutanoic acid}{F <- CH <- CH2 <- CH2 <- C <- O <- H}}\ \end{array}\] |
\[\begin{array}{cc} \ce{CH3}\phantom{..............}\ce{O}\phantom{........}\\ \phantom{.}|\phantom{.................}||\phantom{........}\\ \ce{\underset{3-Fluorobutanoic acid}{F <- CH <- CH2 <- C <- O <- H}}\ \end{array}\] |
The inductive effect decreases with distance, so −I effect of F is stronger in 3-fluorobutanoic acid than in 4-fluorobutanoic acid. Hence, CH3CHFCH2COOH is stronger acid than FCH2CH2CH2COOH.
APPEARS IN
संबंधित प्रश्न
How is 1-nitropropane prepared from suitable oxime?
How are 2 - nitropropane prepared from suitable oxime?
Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Give two reasons.
Why pKa of F-CH2-COOH is lower than that of Cl−CH2−COOH?
Distinguish between the following : Benzoic acid and methyl benzoate
CH3CO2H or CH2FCO2H
Which acid of the pair shown here would you expect to be stronger?
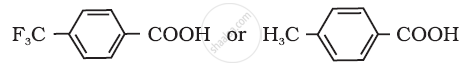
Write the reactions involved Hell-Volhard Zelinsky reaction?
What happens when Salicylic acid is treated with (CH3CO)2 O/H+?
Complete the following reaction sequence.
\[\begin{array}{cc}
\ce{O}\phantom{...............................................}\\
||\phantom{...............................................}\\
\ce{CH3 - C - CH3 ->[(i) CH3MgBr][H2O] (A) ->[Na metal][Ether] (B) ->[CH3 - Br] (C)}
\end{array}\]
Acidity of BF3 can be explained on the basis of which of the following concepts?
When propionamide reacts with Br2 in the presence of alkali the product is ______.
Na2CO3 cannot be used in place of (NH4)2CO3 for the precipitation of the V group because ______.
Describe the action of alcoholic potassium hydroxide (alc. KOH) on ethyl bromide
Describe the action of alcoholic potassium hydroxide (alc. KOH) on isopropyl bromide.
