Advertisements
Advertisements
प्रश्न
Which of the following compounds is most reactive towards nucleophilic addition reactions?
पर्याय
\[\begin{array}{cc}
\phantom{...}\ce{O}\\
\phantom{...}||\\
\phantom{}\ce{CH3 - C - H}
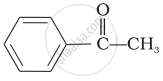
\end{array}\]\[\begin{array}{cc}
\phantom{.}\ce{O}\\
\phantom{.}||\\
\phantom{}\ce{CH3 - C - CH3}
\end{array}\]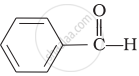
उत्तर
\[\begin{array}{cc}
\phantom{...}\ce{O}\\
\phantom{...}||\\
\phantom{}\ce{CH3 - C - H}
\end{array}\]
Explanation:
\[\ce{CH3CHO}\] is most reactive towards nucleophilic addition reactions. Carbonyl compounds are polar with positive charge on carbon atom which is attacked by nucleophiles. Two electron releasing alkyl groups in ketones make carbonless electron deficient than aldehydes. Benzene ring exhibits + R-effect which thereby decreases the ease of nucleophilic addition reaction in benzaldehyde and acetophenone. Hence the reactivity order is
\[\begin{array}{cc}
\phantom{...}\ce{H}\phantom{..........}\ce{H}\phantom{.........}\ce{R}\phantom{.......}\\
\phantom{......}\backslash\phantom{...........}\backslash\phantom{.........}\backslash\phantom{........}\\
\phantom{.......}{\overset{δ+}{\ce{C}} = \overset{δ-}{\ce{O}} > \phantom{..}\overset{δ+}{\ce{C}} = \overset{δ-}{\ce{O}} > \phantom{..}\overset{δ+}{\ce{C}} = \overset{δ-}{\ce{O}}}\phantom{}\\
\phantom{.......}/\phantom{...........}/\phantom{...........}/\phantom{........}\\
\phantom{.....}\ce{H}\phantom{..........}\ce{R}\phantom{..........}\ce{R}\phantom{.........}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write the products formed when CH3CHO reacts with the following reagents : HCN
What is meant by the following term? Give an example of the reaction in the following case.
Schiff’s base
How will you convert benzoic acid to m-bromobenzoic acid?
Benzaldehyde can be obtained from benzal chloride. Write reactions for obtaining benzalchloride and then benzaldehyde from it.
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
Which of the following has the most acidic hydrogen?
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
The increasing order of the following compounds towards HCN addition is:
| (i) | 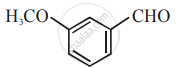 |
| (ii) | 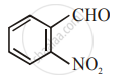 |
| (iii) | 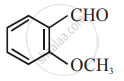 |
| (iv) | 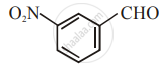 |
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
