Advertisements
Advertisements
Question
Which of the following compounds is most reactive towards nucleophilic addition reactions?
Options
\[\begin{array}{cc}
\phantom{...}\ce{O}\\
\phantom{...}||\\
\phantom{}\ce{CH3 - C - H}
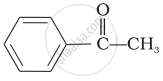
\end{array}\]\[\begin{array}{cc}
\phantom{.}\ce{O}\\
\phantom{.}||\\
\phantom{}\ce{CH3 - C - CH3}
\end{array}\]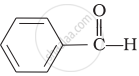
Solution
\[\begin{array}{cc}
\phantom{...}\ce{O}\\
\phantom{...}||\\
\phantom{}\ce{CH3 - C - H}
\end{array}\]
Explanation:
\[\ce{CH3CHO}\] is most reactive towards nucleophilic addition reactions. Carbonyl compounds are polar with positive charge on carbon atom which is attacked by nucleophiles. Two electron releasing alkyl groups in ketones make carbonless electron deficient than aldehydes. Benzene ring exhibits + R-effect which thereby decreases the ease of nucleophilic addition reaction in benzaldehyde and acetophenone. Hence the reactivity order is
\[\begin{array}{cc}
\phantom{...}\ce{H}\phantom{..........}\ce{H}\phantom{.........}\ce{R}\phantom{.......}\\
\phantom{......}\backslash\phantom{...........}\backslash\phantom{.........}\backslash\phantom{........}\\
\phantom{.......}{\overset{δ+}{\ce{C}} = \overset{δ-}{\ce{O}} > \phantom{..}\overset{δ+}{\ce{C}} = \overset{δ-}{\ce{O}} > \phantom{..}\overset{δ+}{\ce{C}} = \overset{δ-}{\ce{O}}}\phantom{}\\
\phantom{.......}/\phantom{...........}/\phantom{...........}/\phantom{........}\\
\phantom{.....}\ce{H}\phantom{..........}\ce{R}\phantom{..........}\ce{R}\phantom{.........}\\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
What is meant by the following term? Give an example of the reaction in the following case.
Imine
How will you bring about the following conversion in not more than two steps?
Bromobenzene to 1-Phenylethanol
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Write balanced chemical equations for action of ammonia on - acetone
Acetone, Acetaldehyde, Benzaldehyde, Acetophenone – reactivity towards addition of HCN.
Alkenes 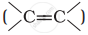 and carbonyl compounds
and carbonyl compounds 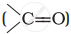 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Reaction of aqueous sodium hydroxide on chlorobenzene gives which of the following products?
What is the action of sodium hypoiodite on acetone?
Which will undergo faster nucleophilic addition reaction?
Acetaldehyde or Propanone
Draw the structure of the following derivative.
The ethylene ketal of hexane-3-one
