Advertisements
Advertisements
Question
The correct order of increasing acidic strength is ______.
Options
Phenol < Ethanol < Chloroacetic acid < Acetic acid
Ethanol < Phenol < Chloroacetic acid < Acetic acid
Ethanol < Phenol < Acetic acid < Chloroacetic acid
Chloroacetic acid < Acetic acid < Phenol < Ethanol
Solution
The correct order of increasing acidic strength is Ethanol < Phenol < Acetic acid < Chloroacetic acid.
Explanation:
Because the phenoxide ion produced is stabilised by resonance, phenol is more acidic than ethanol. When it comes to ethanol, resonance does not help to stabilise the phenoxide ion. The acidity of chloroacetic acid is higher than that of acetic acid. Alcohols and phenols are less acidic than carboxylic acids.
APPEARS IN
RELATED QUESTIONS
Write the structures and IUPAC names of the :Adipic acid compound
Give the IUPAC name of the following compound:
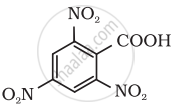
How do you convert the Toluene to Benzoic acid
Identify the compounds A, B, C and D.
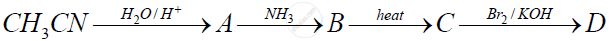
How do you convert the following :
Ethyne to Ethanal
How do you convert the following :
Acetic acid to Methane
Write the equations involved in the following reactions :
Stephen reaction
Write the equations involved in the following reactions :
Wolff-Kishner reduction
Write the equations involved in the following reactions :
Etard reaction
Complete the following:
\[\ce{C6H5NO2 ->[Sn/HCl] A ->[Br2/H2O] B ->[NaNO2/HCl][273 - 278 K] C ->[HBF4][Δ] D}\]
