Advertisements
Advertisements
प्रश्न
Write the products formed when CH3CHO reacts with the following reagents : HCN
उत्तर १
Acetaldehyde (CH3CHO) reacts with hydrogen cyanide HCN to give 2-Hydroxypropapanenitrile as product.

उत्तर २
Acetaldehyde add a molecule of hydrogen to form acetaldehyde cyanohydrin.

APPEARS IN
संबंधित प्रश्न
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
Predict the product of the following reaction:
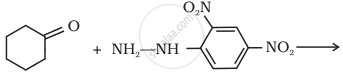
What is meant by the following term? Give an example of the reaction in the following case.
Oxime
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
How are the following compounds prepared?
benzaldehyde from benzoyl chloride
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
A Idol condensation will not be observed in
Paraldehyde is formed as a result of polymerisation:-
The most stable reagent for the conversion of R – CH2OH → RCHO is
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
