Advertisements
Advertisements
प्रश्न
Why are elemental dopants for Silicon or Germanium usually chosen from group XIII or group XV?
उत्तर
The size of dopant atoms should be such as not to distort the pure semiconductor lattice structure and yet easily contribute a charge carrier on forming covalent bonds with Si or Ge.
APPEARS IN
संबंधित प्रश्न
Distinguish between 'intrinsic' and 'extrinsic' semiconductors
In a p-type semiconductor, the acceptor impurity produces an energy level ______
A donor impurity results in ______.
In p-type semiconductor, the dopant is ______.
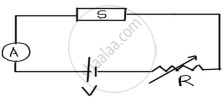
The figure shows a piece of pure semiconductor S in series with a variable resistor R and a source of constant voltage V. Should the value of R be increased or decreased to keep the reading of the ammeter constant, when semiconductor S is heated? Justify your answer
Name the extrinsic semiconductors formed when pure germanium is doped with a trivalent impurity. Draw the energy band diagram of extrinsic semiconductors so formed.
- Statement I: By doping silicon semiconductor with pentavalent material, the electrons density increases.
- Statement II: The n-type semiconductor has net negative charge. In the light of the above statements, choose the most appropriate answer from the options given below:
In an extrinsic semiconductor, the number density of holes is 4 × 1020 m-3. If the number density of intrinsic carriers is 1.2 × 1015 m-3, the number density of electrons in it is ______.
Pieces of copper and of silicon are initially at room temperature. Both are heated to temperature T. The conductivity of ______.
With an increase in the temperature, the electrical conductivity of a semiconductor ______.
