Advertisements
Advertisements
Question
Why are elemental dopants for Silicon or Germanium usually chosen from group XIII or group XV?
Solution
The size of dopant atoms should be such as not to distort the pure semiconductor lattice structure and yet easily contribute a charge carrier on forming covalent bonds with Si or Ge.
APPEARS IN
RELATED QUESTIONS
Semiconductors formed by doping germanium (Ge) with aluminium (Z = 13) and antimony (Z = 51) are ______.
When p-n junction diode is forward biased, then ______.
State how a p-type semiconductor will be obtained from a pure crystal of a semiconductor.
Distinguish between n-type and p-type semiconductors.
The conductivity of a semiconductor increases with increase in temperature because ______.
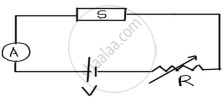
The figure shows a piece of pure semiconductor S in series with a variable resistor R and a source of constant voltage V. Should the value of R be increased or decreased to keep the reading of the ammeter constant, when semiconductor S is heated? Justify your answer
Name the extrinsic semiconductors formed when pure germanium is doped with a trivalent impurity. Draw the energy band diagram of extrinsic semiconductors so formed.
- Assertion (A): Putting the p-type semiconductor slab directly in physical contact with the n-type semiconductor slab cannot form the pn junction.
- Reason (R): The roughness at contact will be much more than inter atomic crystal spacing and continuous flow of charge carriers is not possible.
What type of semiconductor is obtained when a crystal of silicon is doped with a trivalent element?
The majority charge carriers in a P-type semiconductor are ______.
