Advertisements
Advertisements
प्रश्न
Why homologous series of carbon compounds are so called? Write chemical formula of two consecutive members of a homologous series and state the part of these compounds that determines their (i) physical properties, and (ii) chemical properties.
उत्तर
Homologous series of carbon compounds are so called because in such a series of compounds, the same functional group dictates the properties of the carbon compound regardless of the length of the carbon chain. The two consecutive members of a homologous series are CH3OH and C2H5OH (belong to alcohol)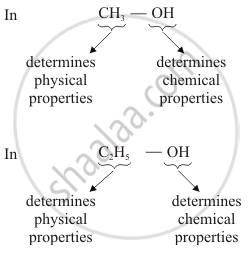
संबंधित प्रश्न
Write the molecular formula of first two members of homologous series having functional group -Cl.
Give the structural formula for 1, 2-dichloroethane
Complete the following table which relates to the homologous series of hydrocarbons.
|
General Formula |
IUPAC name of the homologous series | Characteristic bond type | IUPAC name of the first member of the series |
| `C_nH_(2n-2)` | (A)________ | (B)______ | (C)______ |
| C_nH_(2n+1) | (B)________ | (E)______ | (F)______ |
What is the difference between two consecutive homologues:
(1) in terms of molecular mass?
(2) in terms of number and kind of atoms per molecule?
Give the molecular formula of one homologue of each of the following:
C2H6
Two statements are given - one labeled Assertion (A) and the other labeled Reason (R).
Assertion (A): In a homologous series of alcohols, the formula for the second member is C2H5OH and the third member is C3H7OH.
Reason (R): The difference between the molecular masses of the two consecutive members of a homologous series is 144.
Which of the following does not belong to the same homologous series?
Name the following organic compound:
The first homologue whose general formula is CnH2n.
Consider the carbon compounds having following molecular formula:
(i) C3H6 (ii) C3H8 (iii) C4H6 (iv) C6H6 (v) C6H12
- State the number of double covalent bonds present in C3H6.
- Write the formula of first member of the homologous series to which the carbon compound C4H6 belongs.
- Which one of the above compounds forms a ring structure of carbon atoms?
- Identify, which of the above compounds, is a member of alkane series.
Name the following:
Group of organic compounds where the successive members follow a regular structural pattern, successive compounds differ by a 'CH2' group.
