Advertisements
Advertisements
प्रश्न
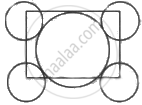
With the help of a labelled diagram show that there are four octahedral voids per unit cell in a cubic close packed structure.
उत्तर
In ccp, each cube consists of eight cubic components, number of atoms per unit cell in ccp is
Where Nc = No. of atoms at corner
Nf = No. of atoms at face centre
= Nc × Nf × contribution
= `8 xx 1/8 + 6 xx 1/2` = 4
Position of octahedral voids = Edge centre and body centre
Number of actahedral voids per unit cell in cubic close packing = `N_e xx 1/4 + N_b xx 1`
= `12 xx 1/4 + 1 xx 1` = 4
⇒ Number of octahedral voids = 4.
APPEARS IN
संबंधित प्रश्न
What is the two dimensional coordination number of a molecule in square close packed layer?
A cubic solid is made of two elements P and Q. Atoms of Q are at the corners of the cube and P at the body-centre. What is the formula of the compound? What are the coordination numbers of P and Q?
Aluminium crystallises in a cubic close-packed structure. Its metallic radius is 125 pm.
(i) What is the length of the side of the unit cell?
(ii) How many unit cells are there in 1.00 cm3 of aluminium?
The coordination number of AB having rock salt geometry is ____________.
In NaCl structure ____________.
How can you best describe the elongated octahedral structure of blue vitriol, CuSO4.5H2O?
In the SF4 molecule, there are:
The packing efficiency of the two dimensional square unit cell shown below is:
Which of the following is frenkal effect.
Element 'B' forms ccp structures and A occupies half of the octahedral voids, while oxygen atoms occupy all the tetrahedral voids. The structure of bimetallic oxide is ______.
