Advertisements
Advertisements
प्रश्न
Show that in a cubic close packed structure, eight tetrahedral voids are present per unit cell.
उत्तर
In ccp structure unit cell is divided into 8 small cubes. Each small cube has atoms at alternative corners [Figure]. In all, each small cube has 4 atoms. When joined to one another, they make a regular tetrahedron. Thus, there is one tetrahedral void in each small cube and 8 tetrahedral void in total. Each of the eight small cubes has one void in one unit cell of ccp structure. We know that ccp structure has four atoms per unit cell. Thus the number of tetrahedral voids is twice the number of atoms.
(a)
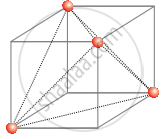
(b)
Again, the number of tetrahedral voids in ccp structure is equal to twice the total number of atoms.
APPEARS IN
संबंधित प्रश्न
How many tetrahedral voids can exist per unit cell in a hexagonal close packing sphere?
The unit cell of a substance has cations A+ at the corners of the unit cell and the anions B− at the center. The simplest formula of the substance is ____________.
Total number of voids in 0.5 mole of a compound which forms hexagonal close packed structure is ____________.
What is the coordination number of sodium in Na2O?
The Ca2+ and F– are located in CaF2 crystal, respectively at face centered cubic lattice points and in ____________.
The number of octahedral voids present in a lattice is A. The number of closed packed articles, the number of tetrahedral voids generated is B the number of closed packed particles:
If Germanium crystallises in the same way as diamond, then which of the following statement is not correct?
In the cubic close packing, the unit cell has ______.
The coordination number of Y will be in the XY types of crystal:
In which of the following structures coordination number for cations and anions in the packed structure will be same?
