Advertisements
Advertisements
प्रश्न
Write chemical reactions for different steps in the manufacture of sulphuric acid by lead chamber process. Draw the structure of phosphorous pentachloride
उत्तर
2SO2 + O2 +2H2O`overset("No" )(->)`2H2SO4
The above net reaction can be written as the sum of the following reactions taking place in different steps.
1) 2H2O +NO `rightarrow` HNO3 +3H+
2) S + O2 `rightarrow` SO2 (Oxidation of sulphur)
3) 2HNO3 + 2SO2 `rightarrow` H2O +NO +NO2 +2SO3
Nitric acid or NO2 is used to oxidize SO2 to SO3.
Or SO2 +NO2 `rightarrow` SO3 + NO
4) SO3 +H2O `rightarrow` H2SO4
Sulphur trioxide reacts with water to form H2SO4 .
Structure of phosphorous pentachloride :
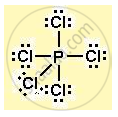
Phosphorous has 5 valence electrons. Each electron is shared with a chlorine atom.
APPEARS IN
संबंधित प्रश्न
What is molecular formula of oleum?
(a) H2SO3
(b) H2SO4
(c) H2S2O7
(d) H2S2O8
Write the conditions to maximize the yield of H2SO4 by contact process.
Complete the following equations: CaF2+H2SO4 →
Complete the following equations: C + conc. H2SO4 →
Draw the structure of the following:
H2SO4
Mention three areas in which H2SO4 plays an important role.
Describe the manufacture of H2SO4 by contact process?
Write balanced equations for NaCl is heated with sulphuric acid in the presence of MnO2
What is the action of concentrated sulphuric acid on phosphorous pentachloride
What happens when dilute sulphuric acid is treated with Fe
What is the action of concentrated sulphuric acid on potassium chlorate?
Write a balanced chemical equation for the following reaction:
Sulphuric acid is treated with phosphorous.
Write structure and molecular formula for the following compounds:
a. Orthophosphoric acid
b. Sulphurous acid
Write the formula for pentaamminechlorocobalt (III) sulphate.
Write the reaction of conc. \[\ce{H2SO4}\] with sugar.
Which of the following reaction is NOT involved in contact process used for manufacturing sulfuric acid?
Identify the INCORRECT statement about H2SO4.
Identify the products formed when conc. sulfuric acid reacts with calcium fluoride.
The molecular formula of oleum is ____________.
The sulfuric acid obtained by contact process is ____________ % pure.
Which among the following catalysts is used in manufacture of sulphuric acid by contact process?
In the manufacture of sulphuric acid by contact process Tyndall box is used to ____________.
The oxidation number of sulphur in Na2S4O6 is:-
