Advertisements
Advertisements
प्रश्न
Write chemical test to distinguish between the following compounds:
Phenol and Benzoic acid
Give chemical test to distinguish between the following pair of compound:
Phenol and Benzoic acid
उत्तर १
Phenol and Benzoic Acid can be distinguished by the iron chloride FeCl3 test. As phenol gives violet colouration with neutral FeCl3, solution while benzoic acid gives buff-coloured precipitate of ferric benzoate.
\[\ce{6C6H5OH + FeCl3 -> [\underset{Violet complex}{Fe(OC6H5)6]^{3-}} + 3H^{+} + 3HCl}\]
\[\ce{\underset{Benzoic acid}{3C6H5COOH} + FeCl3 -> \underset{Buff coloured ppt.}{(C6H5COO)3Fe} + 3HCl}\]
उत्तर २
| Reagent | Phenol | Benzoic acid |
| Neutral ferric chloride | When phenol is treated with ferric chloride, there is a change in colour; the product formed is either green, blue, purple, or red. | No change in the colour of benzoic acid. |
APPEARS IN
संबंधित प्रश्न
Distinguish between: CH3COOH and HCOOH
Give a simple chemical test to distinguish between the following pair of compounds:
Pentan-2-one and Pentan-3-one
Give a simple chemical test to distinguish between the following pair of compounds:
Benzaldehyde and Acetophenone
Complete the synthesis by giving missing starting material, reagent or product.
Which of the following compounds will give butanone on oxidation with alkaline \[\ce{KMnO4}\] solution?
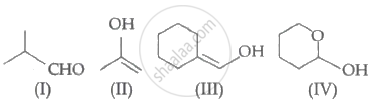
Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
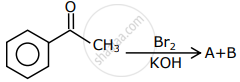
The major products formed in the following reaction sequence A and B are:
An organic compound neither reacts with neutral ferric chloride solution nor with Fehling solution. It however, reacts with Grignard reagent and gives positive iodoform test. The compound is:
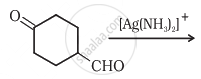
In Tollen's test for aldehyde, the overall number of electrons(s) transferred to the Tollen's reagent formula \[\ce{[Ag(NH3)2]+}\] per aldehyde group to form silver mirror is ______. (Round off to the nearest integer)
The reagent that can be used to distinguish acetophenone and benzophenone is ______.
