Advertisements
Advertisements
प्रश्न
Write the equation taking place at the anode.
उत्तर
Ag - e- → Ag+
Cu - e- → Cu2+
Cl⁻ ‒ e- → Cl
Cl + Cl → Cl2
APPEARS IN
संबंधित प्रश्न
Copy and complete the following table which refers to the conversion of ions to neutral particles.
| Conversion | Ionic equation | Oxidation / Reduction |
| Chloride ion to chlorine molecule | 1)_________ | 2)_________ |
| Lead (II) ion to lead | 3)_________ | 4)_________ |
A solution of HCl gas in water conducts electricity because ______, but a solution of HCl gas in toluene does not conduct an electric current because ______.
Define the term : Non-electrolyte
Write the difference between with examples:
Electrolytic dissociation and ionization
Write the equations for the reactions, which takes place at the electrodes during the electrolysis of lead bromide?
Fill in the blank.
As we descend the electrochemical series containing cations, the tendency of the cations to get ________ at the cathode increases.
Select the correct answer from the choices a,b,c and d which are given. Write only the letter corresponding to the correct answer.
During ionization metals lose electrons, this change can be called _______________.
Fill in the blank from the choices given below :
Electro covalent compounds have a _____ boiling point
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
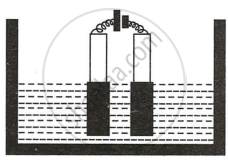
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Give appropriate scientific reasons for the following statement :
The electrical conductivity of acetic acid is less in comparision to the electrical conductivity of dilute sulphuric acid at a given concentration.
