Advertisements
Advertisements
प्रश्न
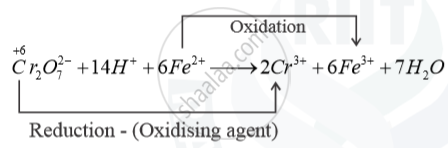
Write the ionic equation showing the oxidation of Fe(II) salt by acidified dichromate solutions.
उत्तर
Oxidation of Fe2+ salt by acidified solution K2Cr2O7 solution
APPEARS IN
संबंधित प्रश्न
Complete the following equations: Cr2O72- + 14H+ + 6I →
Give an example and suggest a reason for the following feature of the transition metal chemistry:
The lowest oxide of transition metal is basic, the highest is amphoteric/acidic.
Complete the following equations:
`2MnO_4^(-)+16H^++5S^(2-)rarr`
E0 value for Mn3+ Mn2+ couple is much more positive than that for Cr3-/ Cr2-.
Generally transition elements and their salts are coloured due to the presence of unpaired electrons in metal ions. Which of the following compounds are coloured?
(i) \[\ce{KMnO4}\]
(ii) \[\ce{Ce(SO4)2}\]
(iii) \[\ce{TiCl}\]
(iv) \[\ce{Cu2 Cl2}\]
The product formed upon heating methyl bromide with potassium tert-butoxide is
KMnO4 is coloured due to ______.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
