Advertisements
Advertisements
प्रश्न
E0 value for Mn3+ Mn2+ couple is much more positive than that for Cr3-/ Cr2-.
उत्तर
Much larger 3rd ionisation energy of Mn (where the required change is d5 to d4) is mainly responsible for this.
Mn3+ = 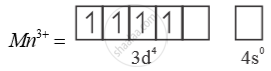
Mn2+ = 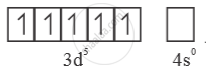 → Half filled (more stable)
→ Half filled (more stable)
APPEARS IN
संबंधित प्रश्न
Complete the following equations:
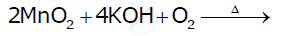
Complete the following equations: Cr2O72- + 14H+ + 6I →
Complete the following chemical equation
8MnO4- + 3S2O32- + H2O →
Complete the following equations : 2 Na2CrO4 + 2 H + →
Indicate the steps in the preparation of K2Cr2O7 from chromite ore.
Which of the following reactions are disproportionation reactions?
(a) \[\ce{Cu^{+} -> Cu^{2+} + Cu}\]
(b) \[\ce{3MnO^{-}4 + 4H^{+} -> 2MnO^{-}4 + MnO2 + 2H2O}\]
(c) \[\ce{2KMnO4 -> K2MnO4 + MnO2 + O2}\]
(d) \[\ce{2MnO^{-}4 + 3Mn^{2+} + 2H2O -> 5MnO2 + 4H^{+}}\]
In the form of dichromate, \[\ce{Cr (VI)}\] is a strong oxidising agent in acidic medium but \[\ce{Mo (VI)}\] in \[\ce{MoO3}\] \[\ce{and W (VI)}\] in \[\ce{WO3}\] are not because:
(i) \[\ce{Cr(VI)}\] is more stable than \[\ce{Mo(VI)}\] and \[\ce{and W(VI)}\].
(ii) \[\ce{Mo(VI)}\] and \[\ce{and W(VI)}\] are more stable than \[\ce{Cr(VI)}\].
(iii) Higher oxidation states of heavier members of group-6 of transition series are more stable.
(iv) Lower oxidation states of heavier members of group-6 of transition series are more stable.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
