Advertisements
Advertisements
Question
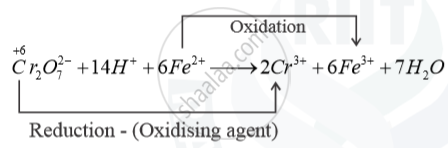
Write the ionic equation showing the oxidation of Fe(II) salt by acidified dichromate solutions.
Solution
Oxidation of Fe2+ salt by acidified solution K2Cr2O7 solution
APPEARS IN
RELATED QUESTIONS
Complete the following equation :
`2MnO_4^(-)+6H^++5NO_2^(-)rarr`
Complete the following equation : MnO4- + 8H+ + 5e- →
Complete the following chemical equations :
Cu + H2SO4(conc.) →
Name a member of the lanthanoid series that is well-known to exhibit +2 oxidation state.
On addition of small amount of \[\ce{KMnO4}\] to concentrated \[\ce{H2SO4}\], a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following.
Which of the following is amphoteric oxide?
\[\ce{Mn2O7, CrO3, Cr2O3, CrO, V2O5, V2O4}\]
Which of the following ions will have a magnetic moment value of 1.73 BM.
\[\ce{Sc^3+, Ti^3+, Ti^2+, Cu^2+, Zn^2+}\]
Indicate the step in the preparation of K2Cr2O7 from chromite ore.
Indicate the steps in the preparation of K2Cr2O7 from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
