Advertisements
Advertisements
प्रश्न
Write one balanced equation to show Emission of `beta^-` (i.e. a negative beta particle)
उत्तर
Emission of `beta^-` (i.e. a negative beta particle)
When an energetic `gamma` - ray photon falls on heavy substance, it is absorbed by some nucleus of the substance, and its energy gives rise to the production of an electron and a position. This phenomenon in which energy is converted into mass is called pair production.
Equation :
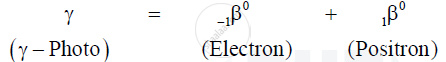
APPEARS IN
संबंधित प्रश्न
Boron has two stable isotopes, `""_5^10"B"` and `""_5^11"B"`. Their respective masses are 10.01294 u and 11.00931 u, and the atomic mass of boron is 10.811 u. Find the abundances of `""_5^10"B"` and `""_5^11"B"`.
The three stable isotopes of neon: `""_10^20"Ne"`, `""_10^21"Ne"` and `""_10^22"Ne"` have respective abundances of 90.51%, 0.27% and 9.22%. The atomic masses of the three isotopes are 19.99 u, 20.99 u and 21.99 u, respectively. Obtain the average atomic mass of neon.
Two nuclei have mass numbers in the ratio 1: 2. What is the ratio of their nuclear densities?
The atomic mass of Uranium `""_92^238"U"` is 238.0508 u, while that of Thorium `""_90^234"Th"` is 234.0436u, and that of Helium `""_2^4"He"` "is 4.0026u. Alpha decay converts `""_92^238"U"` into `""_92^234"Th"` as, shown below:
`""_92^238"U" -> ( ""_90^234"Th" + ""_2^4"He" + "Energy" )`
\[\ce{^197_79Au}\] contains ______.
The nuclei of isotopes of a given element contain the same number of ______.
A nucleus yYx emits one α and two β particles. The resulting nucleus is ______.
Are the nucleons fundamental particles, or do they consist of still smaller parts? One way to find out is to probe a nucleon just as Rutherford probed an atom. What should be the kinetic energy of an electron for it to be able to probe a nucleon? Assume the diameter of a nucleon to be approximately 10–15 m.
A nuclide 1 is said to be the mirror isobar of nuclide 2 if Z1 = N2 and Z2 = N1. (a) What nuclide is a mirror isobar of 1123 Na? (b) Which nuclide out of the two mirror isobars have greater binding energy and why?
Two nuclei have different mass numbers A1 and A2. Are these nuclei necessarily the isotopes of the same element? Explain.
