Advertisements
Advertisements
प्रश्न
Write true or false. Correct the false statement.
Free radicals are negatively charged species.
पर्याय
True
False
उत्तर
Free radicals are negatively charged species.- False
Correct statement:
Free radicals are electrically neutral/uncharged species.
APPEARS IN
संबंधित प्रश्न
Find out the most stable species from the following. Justify
`dot"CH","CH"_3-dot"CH" - "CH"_3,` \[\begin{array}{cc}\ce{CH3 -\dot{C} - CH3}\\
|\phantom{.}\\\phantom{..}\ce{CH3}\end{array}\]
Find out the most stable species from the following. Justify.
`bar"C""H"_3, bar"C""H"_2"Br", bar"C""Br"_3`
Draw a resonance structure of the following:
Benzaldehyde
Draw a resonance structure of the following:
Buta-1,3-diene
Write true or false. Correct the false statement.
Homolytic fission involves the unsymmetrical breaking of a covalent bond.
Draw all the no-bond resonance structures of isopropyl carbocation.
Choose the correct option.
Which of the following statements are true with respect to electronic displacement in a covalent bond?
a. Inductive effect operates through π bond
b. Resonance effect operates through σ bond
c. Inductive effect operates through σ bond
d. Resonance effect operates through π bond
Choose the correct option.
Hyperconjugation involves overlap of ______ orbitals.
The correct IUPAC name of the compound 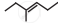 is ______.
is ______.
Choose the correct option.
The geometry of a carbocation is ______.
Choose the correct option.
The delocalization of electrons due to overlap between p orbital and sigma bond is called _______.
Which of the following compound is highly reactive towards HCN?
Which of the following is NOT an electrophile?
Which among the following is a set of nucleophiles?
Which of the following is the most unstable carbocation?
How many tertiary carbon atoms and primary carbon atoms respectively are present in 2-iodo-3, 3- dimethyl pentane?
Which of the following represents a set of nucleophiles?
Which of the following is NOT an electrophile?
Resonance is NOT exhibited by ____________.
How many pi bonds and sigma bonds are present in following molecule?
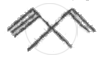
Which of the following statements is not correct?
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
