Advertisements
Advertisements
प्रश्न
Write your observation for the following chemical reaction and name the product formed :
When ammonium chloride is heated with sodium hydroxide.
उत्तर
When solid ammonium chloride is heated with sodium hydroxide solution, a gas ammonia is evolved which is recognised by its strong pungent smell.
\[\ce{\underset{\text{(Solid)}}{Ammonium Chloride}+{Sodium hydroxide}->[heated]\underset{\text{(Solution)}}{Sodium chloride}+Water + \underset{\text{(gas)}}{Ammonia}}\]
APPEARS IN
संबंधित प्रश्न
How will you indicate Exothermic reaction in a chemical equation?
Balance the given equation:
H2O2  H2O + O2
H2O + O2
Balance the given equation:
AI2(SO4)3 +NaOH  AI(OH)3 + Na2SO4
AI(OH)3 + Na2SO4
Balance the following chemical equation:
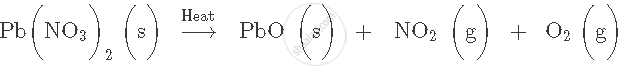
Explain the terms with examples.
Balanced equation
Balance the following equation:
P + HNO3 → NO2 + H2O + H3PO4
Why should an equation be balanced? Explain with the help of a simple equation.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – zinc carbonate.
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
2H2 + O2 → 2H2O
Give the characteristic tests for the following gases :
- CO2
- SO2
- O2
- H2
