Advertisements
Advertisements
Question
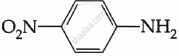
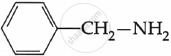
Among the following, which is the strongest base?
Options
Solution
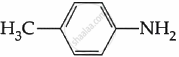
Explanation:
The electron density on the nitrogen atom increases due to the presence of the CH3 group.
APPEARS IN
RELATED QUESTIONS
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
What are amines?
How are amines classified?
\[\ce{Aniline + benzoylchloride ->[NaOH] C6H5 - NH - COC6H5}\] this reaction is known as ____________.
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Arrange the following.
In decreasing order of basic strength in gas phase (C2H5)NH2, (C2H5)NH, (C2H5)3N and NH3.
How will you prepare propan-1-amine from 1-nitropropane?
The following amine can be classified as (C2H5)2NH:
Write a short note on the following
Ammonolysis
Write a short note on the following.
Ammonolysis