Advertisements
Advertisements
Question
An electron in a hydrogen atom makes transitions from orbits of higher energies to orbits of lower energies.
- When will such transitions result in (a) Lyman (b) Balmer series?
- Find the ratio of the longest wavelength in the Lyman series to the shortest wavelength in the Balmer series.
Solution
(i) Emission spectrum of Hydrogen atom: Lyman and Balmer series
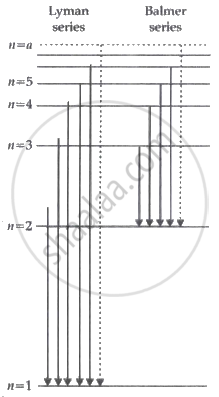
- Lyman Series: When electrons will jump from a higher energy orbit to n = 1 orbit.
- Balmer Series: When electrons will jump from a higher energy orbit yo n = 2 orbits.
(ii) Longest wavelength of Lyman series:
`1/λ_"L" = "R"_"H"[1/1^2 - 1/2^2] = "R"_"H" [3/4]`
Shortest wavelength of Balmer series:
`1/λ_"B" = "R"_"H"[1/2^2 - 1/(∞^2)] = "R"_"H" [1/4]`
Now,
`λ_"L"/λ_"B" = [4/3]/[4/1] = 1/3`
APPEARS IN
RELATED QUESTIONS
The nuclei having same number of protons but different number of neutrons are called ___ .
(a) isobars
(b) a-particles
(c) isotopes
(d) y-particles
When is Hα line in the emission spectrum of hydrogen atom obtained? Calculate the frequency of the photon emitted during this transition.
Find out the wavelength of the electron orbiting in the ground state of hydrogen atom.
The ground state energy of hydrogen atom is −13.6 eV. If and electron make a transition from the energy level −0.85 eV to −3.4 eV, calculate spectrum does his wavelength belong?
A spectroscopic instrument can resolve two nearby wavelengths λ and λ + Δλ if λ/Δλ is smaller than 8000. This is used to study the spectral lines of the Balmer series of hydrogen. Approximately how many lines will be resolved by the instrument?
Name the series in the atomic spectra of the hydrogen atom that falls in the ultra violet region.
When a beam of white light is passed through sodium vapors and then through a spectrometer, spectrum so obtained has two dark lines present in the yellow region. This spectrum is called:
Hydrogen spectrum consists of discrete bright lines in a dark background and it is specifically known as hydrogen emission spectrum. There is one more type of hydrogen spectrum that exists where we get dark lines on the bright background, it is known as absorption spectrum. Balmer found an empirical formula by the observation of a small part of this spectrum and it is represented by
`1/lambda = "R"(1/2^2 - 1/"n"^2)`, where n = 3, 4, 5,....
For Lyman series, the emission is from first state to nth state, for Paschen series, it is from third state to nth state, for Brackett series, it is from fourth state to nth state and for P fund series, it is from fifth state to nth state.
Number of spectral lines in hydrogen atom is ______.
Hydrogen spectrum consists of discrete bright lines in a dark background and it is specifically known as hydrogen emission spectrum. There is one more type of hydrogen spectrum that exists where we get dark lines on the bright background, it is known as the absorption spectrum. Balmer found an empirical formula by the observation of a small part of this spectrum and it is represented by
`1/lambda = "R"(1/2^2 - 1/"n"^2)`, where n = 3, 4, 5,....
For Lyman series, the emission is from first state to nth state, for Paschen series, it is from third state to nth state, for Brackett series, it is from fourth state to nth state and for P fund series, it is from fifth state to nth state.
Which series of hydrogen spectrum corresponds to ultraviolet region?
In the following atoms and molecules for the transition from n = 2 to n = 1, the spectral line of minimum wavelength will be produced by
