Advertisements
Advertisements
Question
Answer the following in brief.
What are valence band and conduction band?
Solution
- Conduction band:
The highest energy band containing electrons is the conduction band. It is formed by the interaction of the outermost energy levels of closely spaced atoms in solids. Conduction band may be partially occupied or vacant.
Electrons in the conduction band are mobile and delocalized over the entire solid. They conduct electricity when an electrical potential is applied. - Valence band:
The band having lower energy than conduction band is the valence band.
The electrons in the valence band are not free to move because they are tightly bound to the respective nuclei.
APPEARS IN
RELATED QUESTIONS
Answer the following in one or two sentences.
The following pictures show population of bands for materials having different electrical properties. Classify them as insulator, semiconductor or metal.
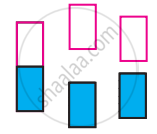
Answer the following in one or two sentences.
How does the electrical conductivity of a semiconductor change with temperature? Why?
Answer the following in one or two sentences.
The picture represents bands of MOs for Si. Label valence band, conduction band, and band gap.
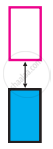
Distinguish with the help of diagrams metal conductors, insulators and semiconductors from each other.
What are n-type semiconductors? Why is the conductivity of doped n-type semiconductor higher than that of pure semiconductor? Explain with diagram.
Explain with diagram, Frenkel defect. What are the conditions for its formation? What is its effect on density and electrical neutrality of the crystal?
Which of the following is a metal?
What is band gap? Arrange band gaps in decreasing order for a semiconductor, metal conductor and insulator.
State the conditions for the formation of Schottky defect. The following picture shows bands for materials having different electrical properties.

Classify them as semiconductors, metals and insulators with reason.
Explain the classification of solids on the basis of electrical conductivity.
