Advertisements
Advertisements
Question
Arrange benzene, n-hexane and ethyne in decreasing order of acidic behaviour. Also give reason for this behaviour.
Solution
Acidic character of a species is defined on the basis of ease with which it can lose its H–atoms.
The hybridization state of carbon in the given compound is:
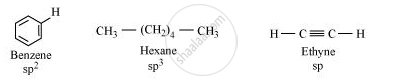
As the s–character increases, the electronegativity of carbon increases and the electrons of C–H bond pair lie closer to the carbon atom. As a result, partial positive charge of H–atom increases and H+ ions are set free.
The s–character increases in the order:
sp3 < sp2 < sp
Hence, the decreasing order of acidic behaviour is Ethyne > Benzene > Hexane
APPEARS IN
RELATED QUESTIONS
Despite their - I effect, halogens are o- and p-directing in haloarenes. Explain.
Suggest a route to prepare ethyl hydrogensulphate (CH3 – CH2 – OSO2 – OH) starting from ethanol (C2H5OH).
Which of the following decolourizes \[\ce{KMnO4}\] (neutral or slightly alkaline)?
Lindlar's catalyst is used for controlled:
An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write IUPAC name of ‘A’.
