Advertisements
Advertisements
Question
Arrange the following as per the instructions given in the brackets:
Cl, F, Br, I (increasing order of electron affinity)
Solution
I < Br < Cl < F
APPEARS IN
RELATED QUESTIONS
Complete the following sentences
Moving across a ………….. of the periodic table the elements show increasing ………………..character (group, period, metallic, non-metallic).
Choose the correct answer.
An alkaline earth metal
(i) Lead
(ii) potassium
(iii) calcium
(iv) Copper
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 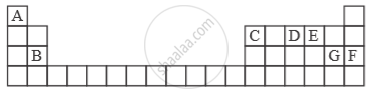
Which non-metallic element has the valency of 2?
State whether the following statement is true or false
All members of zero group are non metals.
Select the correct answer
Which of the following electronic structure is of a metal?
Write the name.
The atom having the smallest atomic radius from zero group.
Write scientific reason.
The non-metallic character increases while going from left to right in a period.
Which of the following metal (s) do not react with water?
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (increasing metallic character)
The given table shows elements with the same number of electrons in its valence shell.
| Elements | A | B | C |
| m.p. | 63.0 | 180.0 | 97.0 |
Arrange them in order of increasing metallic character.
