Advertisements
Advertisements
Question
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Solution

APPEARS IN
RELATED QUESTIONS
Write the reactions involved in the preparation of PVC
Bakelite is the polymer of:
(a) Benzaldchyde and phenol
(b) Acetaldehyde and phenol
(c) Formaldehyde and phenol
(d) Formaldehyde and benzyl alcohol
Based on molecular forces, what type of polymer is neoprene?
Answer the following in one sentence.
Identify 'A' in the following reaction:
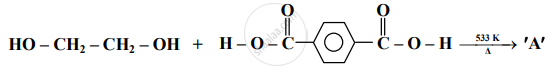
Answer the following in one sentence.
Identify 'B' in the following reaction:
\[\ce{H2N -(CH2)6 - NH2 + HOOC - (CH2)4 - COOH ->[N2][533 K]}\]'B'
Answer the following.
Name the polymer type in which following linkage is present.
\[\begin{array}{cc}\ce{- C - O -}\\||\phantom{.....}\\
\ce{O\phantom{.....}}\end{array}\]
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Answer the following.
Write name and formula of raw material from which bakelite is made.
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Identify condensation polymers and addition polymers from the following.
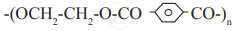
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Answer the following.
Is synthetic rubber better than natural rubber? If so, in what respect?
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Write the name of the catalyst used for preparation of high density polythene polymer.
Which of the following is used as a substitute for wool?
The following structure represents the polymer:
\[\begin{array}{cc}
\ce{[-C-CH2-NH-C-(-CH2)5 NH -]_{{n}}}\\
\phantom{}||\phantom{.............}||\phantom{................}\\
\phantom{}\ce{O}\phantom{.............}\ce{O}\phantom{................}
\end{array}\]
Which among the following polymers is obtained from styrene and 1-3-butadiene?
Identify additional polymers from the following.
I. \[\begin{array}{cc}
\ce{-(CH2 - CH -)_{{n}}}\\
\phantom{....}|\\
\phantom{.......}\ce{C6H5}
\end{array}\]
II. \[\ce{-(CH2 - CH = CH - CH2 -)_{{n}}}\]
III. \[\ce{-(CO(CH2)4 - CONH(CH2)6NH -)_{{n}}}\]
IV.
![]()
Identify the CORRECT statement regarding the following polymer.
\[\begin{array}{cc}
\phantom{....}\ce{O}\phantom{............}\ce{O}\phantom{...................}\ce{H}\phantom{.....}\\
\phantom{....}||\phantom{.............}||\phantom{...................}|\phantom{......}\\
\ce{-[C - (CH2)4 - C - NH - (CH2)6 - N -]_{{n}}}
\end{array}\]
The INCORRECT match for the polymer with its application is:
Identify the INCORRECT match.
Which of the following is the monomer of neoprene?
\[\ce{{n} CH2 = CH2 ->[333 K - 343 K][6 - 7 atm, catalyst] X}\]
Which of the following is CORRECT about polymer 'X'?
Which of the following polymers is a heteropolymer?
Identify the polymer obtained by polymerization of n moles of acrylonitrile.
Which among the following polymers is obtained from CH2 = CH – CN by polymerisation?
Which among the following monomers is used to prepare Teflon?
Which of the following monomers is used in the manufacture of Neoprene rubber?
Which of the following polymer is used to make blankets?
Which among the following polymers is used to manufacture chemical containers?
Which of the following polymers is used as insulation for cables?
Which of the following is not a semisynthetic polymer?
Phenol and formaldehyde undergo condensation to give a polymar (A) which on heating with formaldehyde gives a thermosetting polymer (B). Name the polymers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers?
Which of the following is not a copolymer?
Which of the following polymer has ester linkage?
Trans - form of poly isoprene is:-
Which of the following is an example of polyester?
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
Which of the foolowing polymer is used in the manufacture of insulators.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the name of the monomer of natural rubber.
Match the following pairs:
| Polymer | Monomer | ||
| (i) | Teflon | (a) | CH2 = CH2 |
| (ii) | PVC | (b) | CF2 = CF2 |
| (iii) | Natural rubber | (c) | Isoprene |
| (iv) | Polythene | (d) | H2C=CHCl |
The monomer used in preparation of teflon is ______.
Write the structure and name of monomer of Nylon-6.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
