Advertisements
Advertisements
Question
Boric acid is polymeric due to ______.
Options
its acidic nature
the presence of hydrogen bonds
its monobasic nature
its geometry
Solution
Boric acid is polymeric due to the presence of hydrogen bonds.
Explanation:
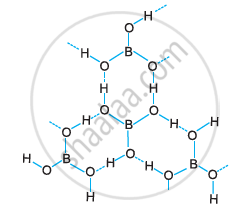
Boric acid is polymeric because of the presence of hydrogen bonds. In the given figure, the dotted lines represent hydrogen bonds.
APPEARS IN
RELATED QUESTIONS
Is boric acid a protic acid? Explain.
Explain what happens when boric acid is heated.
Explain the Structure of Boric Acid.
A certain salt X, gives the following results.
- Its aqueous solution is alkaline to litmus.
- It swells up to a glassy material Y on strong heating.
- When conc. H2SO4 is added to a hot solution of X, white crystal of an acid Z separates out.
Write equations for all the above reactions and identify X, Y and Z.
Write a balanced equation for B2H6 + H2O → ?
An aqueous solution of borax is _______.
Boric acid is an acid because its molecule ______.
When aqueous solution of borax is acidified with hydrochloric acid, a white crystalline solid is formed which is soapy to touch. Is this solid acidic or basic in nature? Explain.
What are boranes? Give chemical equation for the preparation of diborane.
A compound (A) of boron reacts with NMe3 to give an adduct (B) which on hydrolysis gives a compound (C) and hydrogen gas. Compound (C) is an acid. Identify the compounds A, B and C. Give the reactions involved.
Aqueous solution of which of the following boron compounds will be strongly basic in nature?
Boric acid heated to red hot gives ______.
Which of the following compounds are formed when boron trichloride is treated with water?
In diborane ______.
On the addition of mineral acid to an aqueous solution of borax, the compound formed is ______.
Boron reacts with nitric acid to form ______.
The reaction of H3N3B3Cl3(A) with LiBH4 in tetrahydrofuran gives inorganic benzene (B). Further, the reaction of (A) with (C) leads to H3N3B3(Me)3. Compounds (B) and (C) respectively, are ______.
