Advertisements
Advertisements
Question
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb3O4 + HCl ->}\]
Solution
\[\ce{Pb3O4 + 8HCl -> 3PbCl2 +4H2O +Cl2}\]
Concentrated HCl is used.
APPEARS IN
RELATED QUESTIONS
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NaHSO3 + HCl ->}\]
Complete the following reaction and balance them.
Zn+ 2HCl ⟶
Name the following: A greenish yellow gas.
How will you identify?
Chloride ion
How will you identify?
Chlorine gas
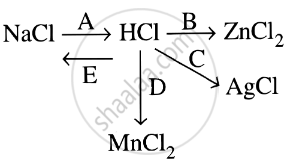
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH +HCI ->}\]
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCI->}\]
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.