Advertisements
Advertisements
Question
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb(NO3)2 + HCl ->}\]
Solution
\[\ce{Pb(NO3)2 +2HCl -> PbCl2 +2HNO3}\]
Dilute HCl is used.
APPEARS IN
RELATED QUESTIONS
Name a chloride which is solube in excess of ammonium hydroxide
Choose the correct answer from the options given below:
Which of the following statement is not correct ?
Name the gas evolved when dilute hydrochloric acid is added to: Lead (II) sulphide
Name the gas evolved when dilute hydrochloric acid is added to: Magnesium bicarbonate
Give a balanced equation when dilute hydrochloric acid is added to : Magnesium bicarbonate
Complete and balance the following reaction, state whether dilutes or conc. acid is used.
\[\ce{NH4OH + HCl -> }\]
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl ->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
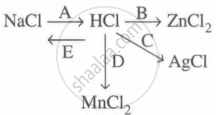
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.