Advertisements
Advertisements
Question
Complete the following equations : 2 Na2CrO4 + 2 H + →
Solution
2 Na2CrO4 + 2 H+ → Na2Cr2O7 + 2 Na+ + H2O
RELATED QUESTIONS
Complete the following equations:
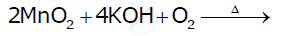
Indicate the steps in the preparation of KMnO4 from pyrolusite ore.
Complete the following equations:
`2MnO_4^(-)+16H^++5S^(2-)rarr`
Using IUPAC norms write the formulae of Potassium trioxalatochromate (III)
\[\ce{KMnO4}\] acts as an oxidising agent in alkaline medium. When alkaline \[\ce{KMnO4}\] is treated with \[\ce{KI}\], iodide ion is oxidised to ______.
The oxidation state of manganese in the product obtained in a reaction of potassium permanganate and hydrogen peroxide in a basic medium is ______.
In the two tetrahedral structures of dichromate ion, ______.
Which of the following ions will have a magnetic moment value of 1.73 BM.
\[\ce{Sc^3+, Ti^3+, Ti^2+, Cu^2+, Zn^2+}\]
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with SO2? Write the ionic equation for the reaction.
