Advertisements
Advertisements
Question
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
Solution
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH] CH2 \underset{\text{Ethylene}}{= CH2 + ZnBr2}}\\
|\phantom{.......}|\phantom{..................................}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{.................................}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify giving reason whether the following compound is aromatic or not.

Identify the compound (A) in the following reaction
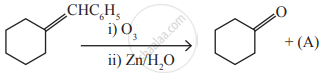
Which of the following is aliphatic saturated hydrocarbon?
Write the chemical equations for combustion of propane.
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
Which of the following is INCORRECT for aromatic hydrocarbons?
Direct bromination of benzene with excess reagent results in the formation of ____________.
Which of the following molecules has shortest C - C bond length?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
